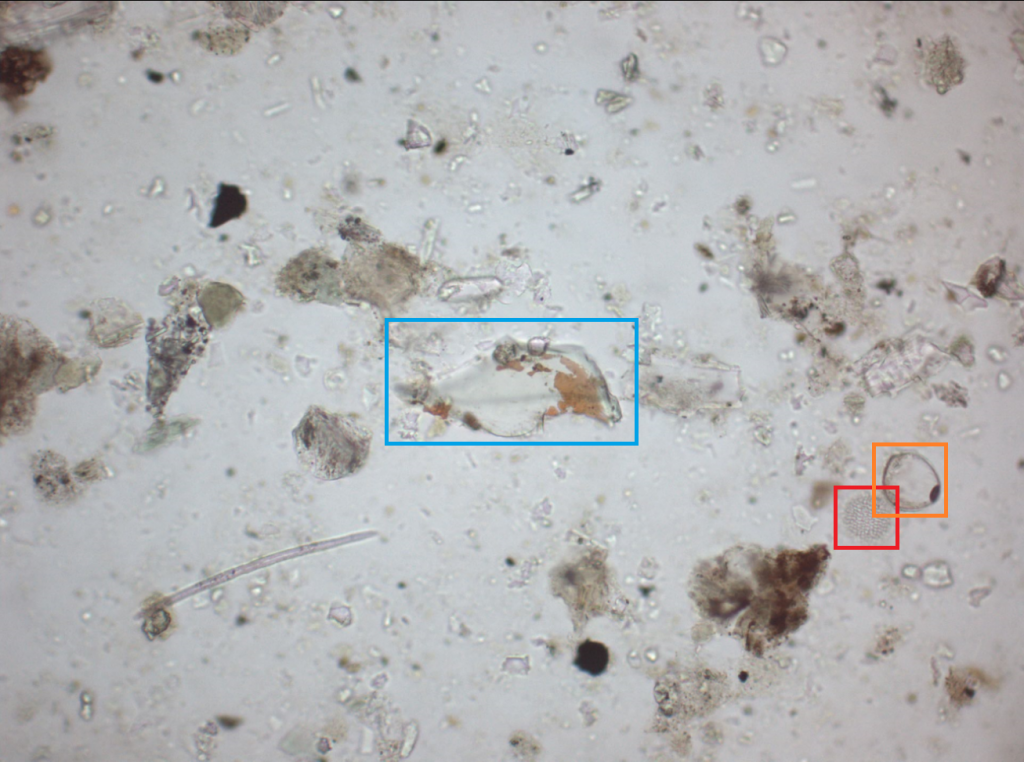
Minerals are found in various shapes and sizes in the deep marine environment. Some minerals form naturally in the ocean through chemical processes, whereas others are transported to the sea from terrestrial environments by wind or water flow. We are going to take a look at a few minerals found at various coring sites in the New Guinea region. With the help of the EDX and knowledge of chemical and physical properties of minerals, at minimum it is possible to determine a minerals group.
At first glance mineral identification on a SEM seems like an impossible task. However, we can deduce that this mineral likely belongs to the amphibole group. This is largely due to its orthorhombic shape and 120° cleavage plane. In mineralogy, cleavage is used to describe how a crystal breaks on a particular plane. EDX point scan analysis indicates that the mineral contains approximately 3% Al, 7% Fe, 8% Mg, 9% Ca, 24% Si, and 41% O. Since the chemical composition of an amphibole is NaCa2(Mg,Fe,Al)5(Al,Si)8O22(OH)2, we can be certain that this mineral is an amphibole.
Is that really gold settling on top of marine sediment? Unfortunately, marine sediments are not a lucrative source of gold. What we are looking at here is pyrite (blue), known as fool’s gold. Pyrite has a chemical composition of FeS2 and its formation is largely dependent on sulfate availability in the ocean, where sulfate is converted by bacteria to hydrogen sulfide. Pyrite commonly forms perfect cubes, however in this case they are visualized as spherical aggregates of microcrystals, called framboids.
Heavy element minerals are indicators of mature, stable sediments. Since heavy minerals are often found concentrated in or around bedrock material, it is likely that this material is terrigenous. EDX microanalysis shows that this mineral contains approximately 17% Si, 50% Zr, and most of the remainder in oxygen. Based on the analysis, it can be inferred that this mineral displaying conchoidal fracture is a zircon (ZrSiO4) crystal.